
Home » Mauna Kea Gains 510(k) Clearance for Two Cellvizio Probes
Mauna Kea Gains 510(k) Clearance for Two Cellvizio Probes
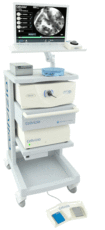
The FDA has granted 510(k) clearance to Mauna Kea Technologies’ Cellvizio Uroflex B and Cystoflex F biopsy probes for use in endo-urology procedures and flexible cystoscopy, respectively, the French devicemaker said Wednesday. The products fall within the company’s flagship Cellvizio line.
The optical biopsy probes are used in situations where traditional biopsies tend to be difficult and ineffective, the company said. Uroflex B assesses the quality of transurethral resection of bladder tumor rigid cystoscopy procedures and may alleviate the risk often seen in such procedures of leaving bladder cancer cells behind, said Mauna Kea spokesman Eric Cohen. The probe also helps physicians better identify the nature of lesions and orient patient treatments, he added.
Cellvizio procedures involve inserting a probe into the working channel of an endoscope. The probe, which appears on the endoscope image, is then placed in contact with mucosa and a real-time video is recorded, the company said.
The company is currently considering options for commercializing the two probes. — Lena Freund
Subscribe to Devices & Diagnostics Letter for complete coverage of the medical devices industry. Click here for more information.
Upcoming Events
-
25Apr
-
07May
-
14May
-
30May
