
Home » CryoLife to Begin Enrollment in Trial of PerClot
CryoLife to Begin Enrollment in Trial of PerClot
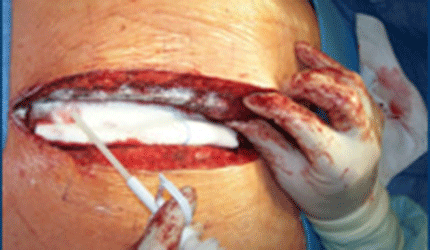
Atlanta devicemaker CryoLife says it will begin enrolling patients in a pivotal clinical trial of its PerClot surgical hemostatic device during the current quarter, following FDA approval of the trial plan.
The prospective, multicenter, multidisciplinary controlled trial will compare PerClot’s safety and efficacy with C.R. Bard’s Arista MPH hemostat as an adjunct to pressure and ligature in achieving hemostasis at two minutes and five minutes. The trial’s safety endpoints are reoperation due to bleeding, hospitalization and procedure time, and complications or adverse events at three month follow-up.
A total of 320 patients from across urological, general and cardiac surgery fields will participate in the study.
PerClot is composed of biocompatible, nonpyrogenic granules derived from plant starch, and delivery applicators. The granules are capable of absorbing water quickly, forming a gel adhesive matrix that stops bleeding and accumulates platelets, red blood cells and clotting proteins, according to CryoLife.
The devicemaker estimates that the U.S. and European hemostatic markets will be worth $915 million and $468 million, respectively, by 2015. PerClot is CE-marked in Europe and available there in 1 g, 3 g and 5 g sizes, with 100 mm or 200 mm applicators. — Lena Freund
Subscribe to Devices & Diagnostics Letter for complete coverage of the medical devices industry. Click here for more information.
Upcoming Events
-
25Apr
-
07May
-
14May
-
30May
