
Home » Hologic Receives Expanded FDA Clearance for Cynosure's SculpSure
Hologic Receives Expanded FDA Clearance for Cynosure's SculpSure
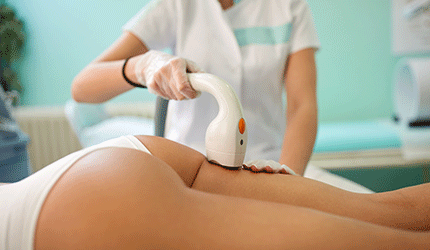
FDA has expanded Hologic’s market clearance for Cynosure's SculpSure to include treatment of the back and inner and outer thighs.
SculpSure utilizes a selective wavelength laser that targets fat cells under the skin. The laser raises the temperature of body fat to disrupt and destroy these cells, which are naturally eliminated over time and do not return. — Cynthia Jessup
Upcoming Events
-
25Apr
-
07May
-
14May
-
30May

