
Home » FDA Clears Siemens Healthineers’ Magnetom Vida 3T MRI System
FDA Clears Siemens Healthineers’ Magnetom Vida 3T MRI System
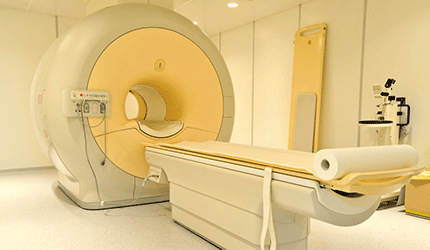
Siemens Healthineers has received FDA market clearance for its Magnetom Vida 3 Tesla (3T) magnetic resonance imaging (MRI) scanner.
Equipped with BioMatrix technology, this scanner can lower the number of rescans and increase productivity to improve MRI’s cost efficiency.
BioMatrix Sensors are built into the scanner’s patient table to automatically track respiratory patterns.
Also, the MRI scanner has BioMatrix tuners which improve the quality and reproducibility of whole-spine diffusion imaging via individual slice adjustments that mitigate distortion.
Lastly, the scanner includes BioMatrix spine coils that acquire and display patient respiratory data without requiring user interaction. — Cynthia Jessup
Upcoming Events
-
25Apr
-
07May
-
14May
-
30May

