
Home » FDA Expects IV Saline Shortage to Improve by Year’s End, Shifts Focus to Amino Acids
FDA Expects IV Saline Shortage to Improve by Year’s End, Shifts Focus to Amino Acids
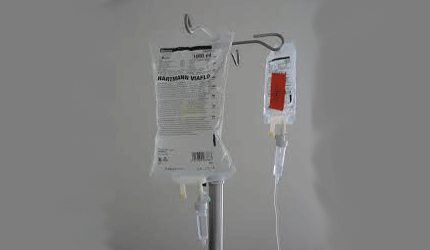
The FDA remains seriously concerned about drug shortages as manufacturers in Puerto Rico continue their slow recovery from Hurricane Maria, but the shortage of IV saline product should improve by the end of the year, Commissioner Scott Gottlieb said in his latest update.
Gottlieb said there are ongoing concerns about other drug shortages — particularly amino acids for injection, used by patients who need intravenous nutrition.
“Like with saline, an ongoing amino acid short supply situation was worsened by Hurricane Maria’s impact on Puerto Rican drug manufacturing facilities,” he said.
The FDA is working with Baxter to address the shortage of IV saline products to stabilize production in its Puerto Rican facilities, and it also approved IV solution products manufactured by Fresenius Kabi and Laboratorios Grifols.
Upcoming Events
-
23Apr
-
25Apr
-
07May
-
14May
-
30May

