
Home » FDA Grants Emergency Use Authorization for bioMérieux’s COVID-19 Test
FDA Grants Emergency Use Authorization for bioMérieux’s COVID-19 Test
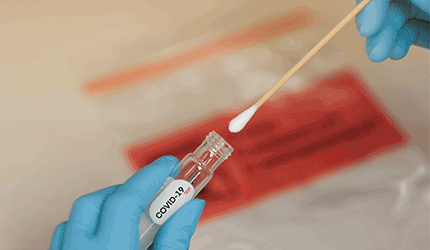
BioMérieux has received an emergency use authorization from the FDA for its Biofire COVID-19 diagnostic for use in certain clinical laboratories.
The EUA allows the French devicemaker’s test to be used for detecting SARS-CoV-2 in moderate and high complexity laboratories.
The test detects the virus in approximately 45 minutes using a nasopharyngeal swab. It is run on the company’s automated Filmarray 2.0 and Filmarray Torch systems.
Upcoming Events
-
25Apr
-
07May
-
14May
-
30May

