www.fdanews.com/articles/179493-fda-approves-cardiac-dimensions-trial-for-heart-valve-repair-device
FDA Approves Cardiac Dimensions Trial for Heart Valve Repair Device
December 1, 2016
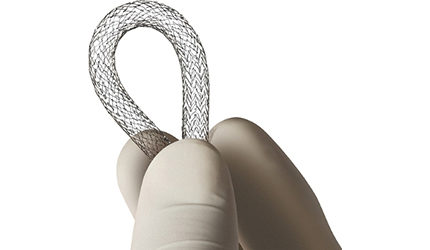
The FDA granted Cardiac Dimensions an investigational device exemption for a pivotal trial of its Carillon heart valve repair device.
The device is a wire with two hooks on each end that is inserted in the heart valve to reshape it by bending the vessel to redirect the blood flow in the heart.
The device met it’s endpoint in the three previous trials done in the EU. — Cynthia Jessup

