
Home » Alere Gets FDA Waiver for Rapid HIV Test
Alere Gets FDA Waiver for Rapid HIV Test
December 12, 2014
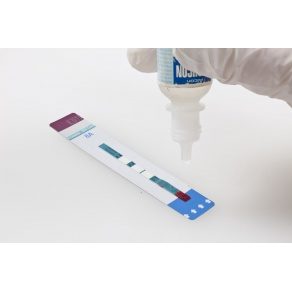
The FDA has granted a waiver under the Clinical Laboratory Improvement Amendments for expanded use of the Alere Determine HIV-1/2 Ag/Ab Combo test for HIV infection. The point-of-care rapid diagnostic test, previously only available to hospitals and laboratories licensed to conduct moderate complexity tests, can now be used in doctor’s offices, clinics and public health settings.
The test, first approved in August 2013, is capable of detecting HIV-1 and HIV-2 antibodies and free HIV p24 antigen, which appears just days after infection and before the HIV antibody is detectable. This helps healthcare providers detect HIV infection earlier in the course of the disease, which in turn can improve clinical outcomes and patient treatment, the Waltham, Mass.-based Alere says.
The Centers for Disease Control and Prevention estimates that 1.1 million people in the U.S. are living with HIV, 15.8 percent of whom have not been diagnosed. — Kellen Owings
Subscribe to Devices & Diagnostics Letter for complete coverage of the medical devices industry. Click here for more information.
Upcoming Events
-
21Oct
