
Home » dorsaVi’s ViMove Wearable Device Clears 510(k) Hurdle
dorsaVi’s ViMove Wearable Device Clears 510(k) Hurdle
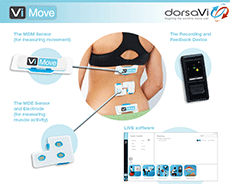
Aussie devicemaker dorsaVi has launched its ViMove wearable monitoring device in the U.S., following the receipt of FDA 510(k) clearance. The device measures and records movement and muscle activity in the lower back and lumbar spine.
The system also measures range of motion in the sagittal and coronal anatomical planes, the Melbourne-based company said Tuesday, adding that it will aid doctors in developing patient treatment plans.
With ViMove, wearable sensors are placed on the body, and patients are assessed as they go through everyday movements and exercises. The device provides instant biofeedback and a concise report for the patient and clinician.
dorsaVi said the launch allows the company to tap into a market of 280,000 physical therapists and 660,000 physicians.
John Kowalczyk, dorsaVi’s U.S. president, said the company will be growing its sales force over the next few weeks to meet anticipated demand for ViMove. — Kellen Owings
Subscribe to Devices & Diagnostics Letter for complete coverage of the medical devices industry. Click here for more information.
Upcoming Events
-
07May
-
14May
-
30May
