
Home » FDA to Mandate Stronger Heart Attack and Stroke Warnings on NSAID Labels
FDA to Mandate Stronger Heart Attack and Stroke Warnings on NSAID Labels
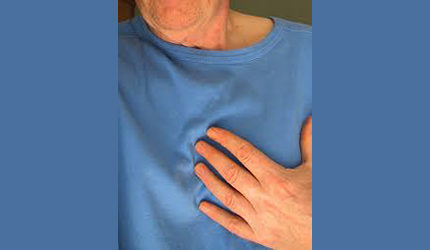
The FDA is requiring stronger label warnings for non-aspirin nonsteroidal anti-inflammatory drugs, after a comprehensive review of new safety information affirmed an increased risk of heart attack or stroke.
While labels for prescription and over-the-counter NSAIDs currently contain information on heart attack and stroke, updates will be required to reflect a litany of new warnings, including that these risks can occur as early as the first weeks of NSAID use and that they increase with longer use and higher doses.
Among other findings, the review suggests that the risk for heart attack or stroke may differ among NSAIDs, the FDA says. Studies also show that risks increase in patients regardless of whether they have heart disease or other risk factors, and that there is a greater risk of heart failure with NSAID use, the FDA says.
The FDA originally described the risks associated with NSAIDs in boxed warnings on drug labels. However, during a February joint meeting of the Arthritis Advisory Committee and the Drug Safety and Risk Management Advisory Committee, advisors pushed for even tougher labeling.
Eighteen companies market NSAIDs in the U.S., including Pfizer, Bayer and Roche.
Read the FDA safety alert at www.fdanews.com/07-13-15-NSAIDLabels.pdf. — Kellen Owings
Upcoming Events
-
07May
-
14May
-
30May
