
Home » FDA Grants Marketing Clearance to SalutarisMD’s RBS System
FDA Grants Marketing Clearance to SalutarisMD’s RBS System
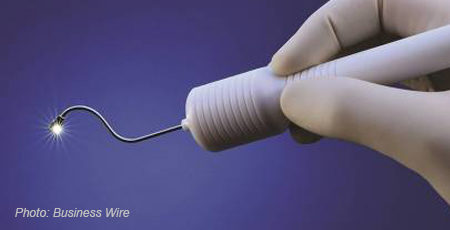
Tucson, Arizona-based Salutaris Medical Devices, Inc. has received marketing clearance for its SMD-Sr90-DA Radionuclide Brachytherapy Source (RBS), which is used within a manual brachytherapy applicator system.
The SMD-Sr90-DA RBS device is indicated for episcleral brachytherapy.
The patented technology delivers a single-use brachytherapy procedure. — Cynthia Jessup
Upcoming Events
-
07May
-
14May
-
30May

